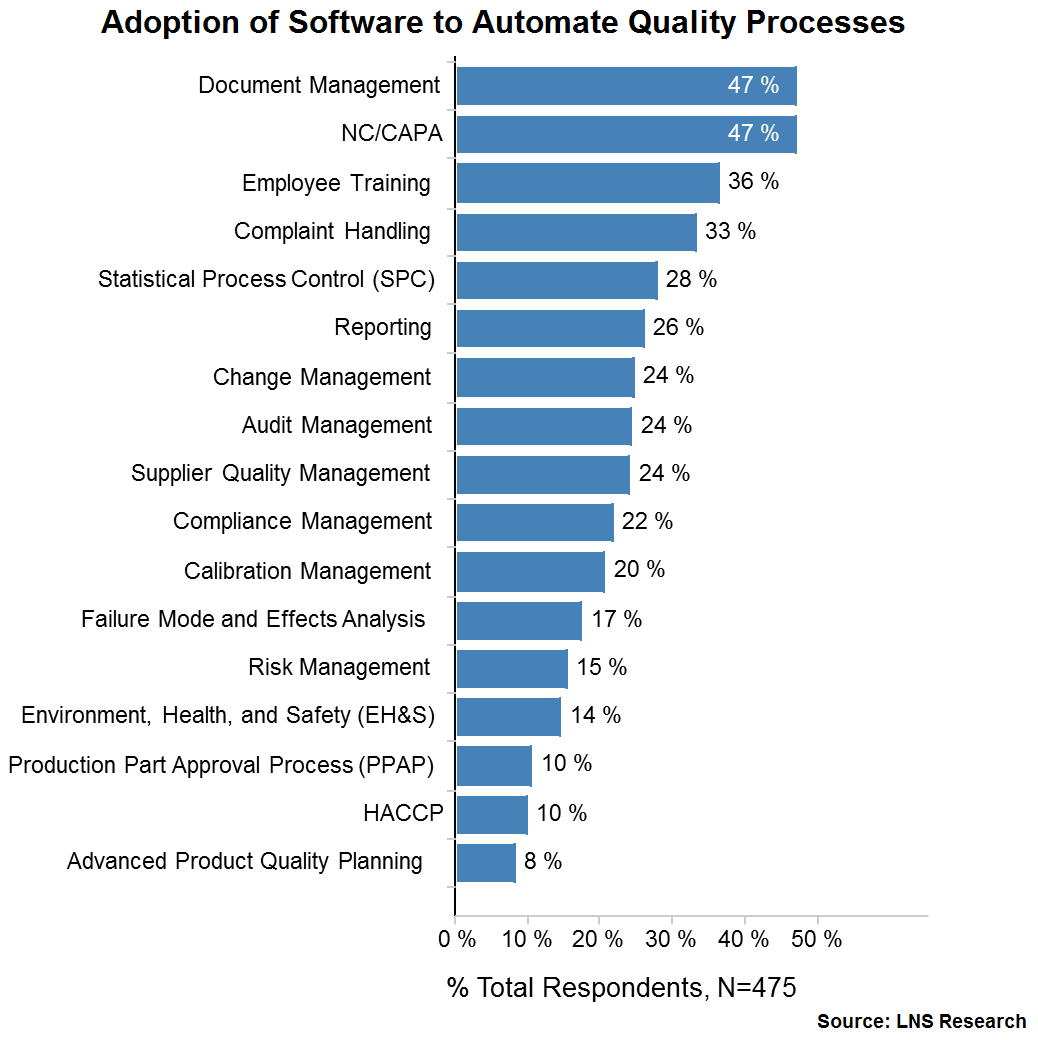
Quality professionals recognize that conformance and harmonization of an enterprise Quality Management System (QMS) begins with a solid document management foundation. But the reality is, most organizations are drowning in documentation—which is why there’s no surprise that document management remains at the very top of quality management processes that have been automated by organizations.
Such capabilities in enterprise quality management software (EQMS) are commonplace, often tightly coupled with training and competence feature-sets, providing an escape from the stagnant and difficult-to-maintain multiple binder, shared directory or 1-dimensional intranet page approach.
Often, it is the case that implementing a document management solution without consideration of overlap with/from additional processes, disciplines (e.g. EHS), upstream and downstream value chain and even the organization as a whole can mean potentially ignoring a huge opportunity for improvement. Quality leaders have a responsibility to provide the most efficient and effective solution for managing this challenging and often difficult-to-tame beast.
In this post, we’re going to take a look at core functionality and considerations that will help an organization evaluate functionality and get the most from its existing or planned deployment. Four primary considerations are below.
1. Speed and ease of access and consumption for document stakeholders
This is the most pervasive problem with document repositories—finding the right document at the right time in an efficient manner. Everyone has anecdotes a plenty of trawling (or leafing) through a repository to find a policy, procedure or work instruction, form or other document. This is wasted time, causes frustration and results in the need for detective skills and likely incurs more time from another resource or two. Looking for documents is an expensive (and frustrating) way for our workforce to spend their time—period.
A one-stop, keyword and advanced search mechanism across all documents (and ideally attachments, too) is one agent of efficiency. Our workforce is used to (and deserves) search engine capability and EQMS providers have made great strides to embed the features in their applications. The application should provide an easy-to-digest format—this can be native through rich text editors and the ability to embed or construct process flow diagrams, or at the very least connect and/or upload version controlled attachments.
Maintaining a consistent and easy-to-read format is crucial to success and this should be a best practice maintained organization-wide. Standard document templates are key here so look for solutions with this capability. We must also consider our procedures and other documents' formatting and readability from the mobile device user’s perspective.
2. Ensuring the integrity of documentation
Having retrieved the desired document quickly, the user has to discern in a rapid manner that it is the most recently published version. A well-designed format will provide this at-a-glance demand and if a previous version of a document is accessed (via search, perhaps for reference), this must be clearly denoted and one simple action should navigate to the latest approved ‘live’ version.
Behind the scenes, document owners and approvers will rely on the ability to work new versions and updates without impacting in-flight documents.
With compliance or costly mistakes on the line, the following must be clear, robust and elegant:
- Version control: can you use your own, guarantee uniqueness and auto-increment?
- Change tracking: what was changed, by whom and when and from/to?
- Approval routing: does it have preconfigured flows, delegation capable, multiple approvers, publication gatekeeper (e-signatures)?
With the increasing affordability and pervasiveness of mobile technology, the need for hard copy documents is diminishing but simple requirements like marking printed documents as being uncontrolled remain important for the time being.
3. Automating onerous tasks associated with publishing and distributing documents
With the peace of mind of knowing documents are robustly ‘controlled,’ an organization must aim to satisfy efficiencies in the final task of publishing, distributing and, where required, driving training requirements, tracking procedure update acknowledgments and training completion.
A review cycle is important for all documents—especially key standard operating procedures. An EQMS solution is by design best placed to achieve a set-and-run approach with minimal overhead. The owner (or owners) of a document or process is known either by name, role or both. By setting a simple recurrence trigger against the document and owner, email and application notification of tasks can be activated and tracked to completion in the document. Without a document management framework this can be onerous to track, error prone and lacking in visibility.
The properties of all documents in a document management component of an EQMS should comprise of metadata, in addition to actual content. This metadata includes but is not limited to:
- Hierarchy of the document; corporate, business unit, region, plant or department
- Distribution by role, function or other element that determines who should need to see access it
- Categorizations based on the scope and types of documentation maintained therein
Triangulating users with roles, responsibilities and document access/requirements facilitates the provision of a user’s personal document library and ideally training and/or competence tracking views. Often this needs to be augmented with capabilities to allow for collaboration across multiple writers, editors, and reviewers both internal and external to the organization. In many cases, if the number of collaborators grows beyond just a few, the editing tools within standard tools like Microsoft Office 365 or Google Docs becomes insufficient.
The configuration and use of organization structure (hierarchy), roles and other metadata should be centralized for consistency and efficiency but also must allow for multiple tiers so that individual facilities and departments can associate documents based on appropriate nuances without disrupting the organization’s approach.
An example of these properties working are a policy and associated form or procedure relating to complaints for a line of business (LOB) in North America that relates only to packaging supervisors. By checking four or five property boxes on the policy and the form, the distribution is set to only those users associated with the matching LOB, geographical context and specific roles. This discrete group can be notified of the new documents and ideally acknowledge them either directly in the application or, as is becoming more common, by accessing AND subsequently responding to the email notifying them of the new or updated document(s). This information is subsequently logged for traceability and later interrogation.
4. Leveraging for multiple disciplines as a business operating system
The features we have discussed thus far are not necessarily specific to quality management. Document management, despite its functional complexity is one of the simplest enterprise management system components to integrate across disciplines. EHS & sustainability has the same demands for policies, procedures, forms, best practices etc. It makes sense to leverage functionality across disciplines like those mentioned but it can and should be considered in an even wider context.
Provided the solution has the appropriate organizational hierarchy mechanism, access control and role-based meta tags, the entire organization can potentially align with the quality management best practices for their document control needs. In this event the solution is elevated to a business operating system component rather than being limited to EQMS. Examples of this can include the management of Standard Operating Procedures (SOPs)—often these impact quality and compliance with standards like ISO but the authoring of SOPs maybe comes from manufacturing or quality engineers and the distribution of SOPs have to go throughout the organization, all the way through to supervisors and operators on the shop floor.
Find the right solution for both the consumers and owners of your EQMS documents
We have discussed some core considerations for document management as a component of your EQMS and potentially beyond. Saving time and equipping the workforce with the right documentation quickly and with confidence will reduce cost and frustration while improving productivity and ensuring compliance. Those responsible for maintaining documentation will yield significant benefits, too, provided they are empowered with the right tools.