Software validations serve an important purpose in life sciences– ensuring that software being used to maintain Part 11 records do so with accuracy, reliability, integrity, availability, and authenticity. However, life science companies validating software against 21-CFR-Part 11 are aware of how resource intensive validation can be.
Click here to speak to Dan
Software validation has historically forced tradeoffs. Life Sciences companies may delay software upgrades for years because of the time and expense of validation, and in the process they may lock themselves out of years of value-adding enhancements. Companies may avoid making certain customizations to avoid the expense of both the initial validation, as well as revalidation upon upgrades; preventing them from unlocking the full potential of adopted software. LNS Survey data supports this: Validation is one of the top challenges in speeding products from R&D to market in Life Sciences.
Figure 1: Top Challenges in Speeding Products from R&D to Market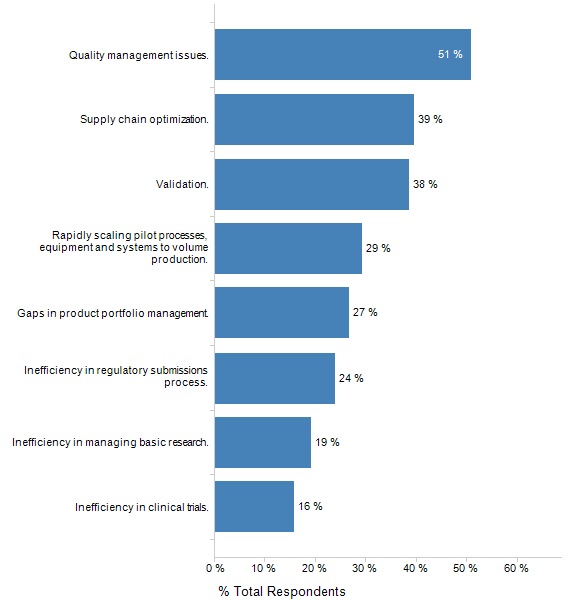
LNS recently published our updated Enterprise Quality Management System Solution Selection Guide and Asset Performance Management Solution Selection Guide; with our solution selection guides for Environmental, Health & Safety (EHS), Manufacturing Operations Management, and our first-ever Industrial Internet of Things (IIoT) Solution Selection Guide on the horizon.
While LNS has been building these solution selection guides, we investigated vendor validation approaches. The good news for life science companies is that the software industry is aware of these challenges, and as a whole is doing a better job of supporting validations. Some vendors who truly understand the challenges are making big strides. These vendors have developed approaches to validation that can greatly reduce the effort involved in validations, and have done so in a manner that has been accepted by the FDA.
Let’s look at how industry is working toward lower effort and FDA-accepted validations.
Validation Documentation: The Industry Baseline
Many vendors claim to have life science industry expertise. Here’s a Go/No Go gauge: do they have pre-built validation documentation? If not, life science is simply not an important industry for them. Validation documentation should be considered a basic expectation for any vendor looking to do more than occasional business in life sciences, unless their offering is highly customized for each customer. Level of completeness can vary, but all vendors should at least provide manual test scripts and expected results. Some provide more complete documentation including full traceability from the expected use requirements to test cases to expected test results.
Automated Validation
Some vendors provide automated validation scripts, which automatically perform validation testing using software testing tools. This approach can deliver substantial time savings. Rather than manually executing complex test scripts for each upgrade, life science companies can execute tests at the click of a button. Of course, the real value isn’t time savings, it’s the impact that these time savings have on the trade-offs we discussed earlier.
When considering a vendor’s automated test offering, determine if they can be modified to accommodate configurations or customizations, consider the resources it will take to make these modifications, consider how the modifications migrate across software versions, and consider how complete the automated validation offering is. Of course, any automation helps, and a risk-based validation approach can help defray any work on modifications.
The Real Difference is Cloud
Cloud-based technologies create new opportunities to streamline validation. Through Cloud, industry leading vendors are providing pre-validated platforms, pre-validated functions, and pre-validated pre-configurations.
There are several vendors that have invested in developing near-zero effort or even true no-effort validation environments. One approach comes from a multi-tenant SaaS (Software as a Service) platform that pre-validates its platform and completely avoids customizations and configurations through options management. Every customer is automatically validated to every update. Other approaches to near-zero effort validations are being developed and have yet to be released; be sure to read about them later this year.
Validation Services
Services can be an important part of the overall validation effort. For instance, validation tools are based on preconfigured processes, and will take some effort to update for customer-specific configurations and customizations, as well as to update between releases. Depending upon the vendor, the documentation updates may be left to the customer, may be performed by the vendor, or may be outsourced to a third party.
This also applies to the actual validation itself. In some cases, the vendor packages the configuration services and validation services and can commit to an established go live timeframe. Many service providers also advocate risk-based validations, which reduce validation time and effort.
Takeaways
Life science companies should prioritize validation support when considering potential software vendors. Think about your ability to leverage new technology not just during the initial purchase, but for years after that purchase. Validations play an important role in a company’s technology lifecycle.
Vendors targeting life science industries should continue to push the boundaries in validation support. Undervaluing the importance of validation can decrease the long term competiveness of both the vendor and its customer base.
NEW eBook is a must for Quality leaders! This eBook provides senior leaders with best practices for building a business case around quality and the foundational framework needed to gain executive commitment.