Regardless of where you look across manufacturing industries, you’re likely to see that the knob on operational speed has been steadily turned up over the past decade. Quality organizations with one foot stuck in the operational past are certainly feeling the pressure to upgrade to integrated quality systems. But the path to successful implementation is frought with challenges from step one.
Increased automation, mobility, Cloud solutions, the Internet of Things (IoT), enterprise manufacturing solutions, etc.—as these trends and technologies take hold, manufacturers are able to collaborate more effectively, forecast and respond to demand more accurately, gain better visibility into internal and external processes, and measure their performance more precisely.
What this means for the quality professional is that that bar for product quality is making bigger and bigger jumps upward, and today is higher than ever. The speed of manufacturing, combined with increasingly global and disparate suppliers, tightening regulations and compliance initiatives, and a sword hanging over the organization’s head in the form of brand damage via social media have placed enormous pressure on quality organization. And more and more, the processes and systems of yesteryear are no longer making the grade.
Research bears this out. Today’s leading organizations are moving beyond manual processes and disparate systems, implementing Enterprise Quality Management Software (EQMS) solutions to integrate and standardize various quality functions at an increasing rate.
According to LNS Research’s quality management study, based on more than 1,200 global quality executives, 32% of respondents have had an EQMS budget that’s increased by more than 20% over the past year, and another 38% of respondents’ budgets have increased by 5% to 10%. It’s clear that EQMS is making its way from the “nice-to-have” category into the “necessary-for-success” realm.
Unfortunately, deriving value from EQMS is not as simple as pasting a dozen vendor logos on a dartboard and letting a random toss decide. It takes time, effort, and all hands on deck.
The Importance of Selection
In order to realize the full value of EQMS imagined at the onset, it’s crucial that you perform due diligence in selecting the solution that will best fit your organization. Many organizations that fail to really hone in on the perfect fit find themselves stalled out in comparison to their initial plans. This may be in the form of fewer rolled-out sites than hoped, or in standardizing fewer quality functions—for instance, starting and stopping at CAPA—than the initial plan called for. So how do you ensure you don’t fall into this trap? You can start with the four broad points outlined below:
Narrowly Define the EQMS Scope
In accurately defining the scope for your EQMS project there are a few important things needed to get started. The starting point for any organization should be aligning with corporate business objectives, which may comprise things like:
- Improving specific metrics (e.g. first pass yield)
- Accelerating the speed of new product introductions
- Reducing customer complaints/returns
- Improved tracking and supplier quality inbound
Understand Delivery Models and Where These Are Changing
As of now data shows that most organizations are still gravitating toward on-premise EQMS solutions, particularly in heavily regulated industries like life sciences and A&D. However, this tide is beginning to turn as advancements in Software as a Service (SaaS), or Cloud technology, is rapidly advancing to address core concerns among users like data security and ubiquitous connectivity while presenting them with hard-to-ignore benefits like lower initial costs and overhead and automatic updates, carrots that are particularly attractive for SMBs that may be IT resource constrained. Does this mean that Cloud is the right choice for you? Not necessarily, but it’s important to understand current trends and take a long view of future desired capabilities as they relate. Depending on your situation, Cloud may be a viable option.
Assemble a Cross Functional Team and Build an RFP
In the RFP process, it’s crucial to organize a cross-functional team of key stakeholders both to begin the decision making process toward an EQMS selection, but more immediately, to accurately determine your current capabilities. This should include team members from groups such as IT, quality executives, plant managers, plant quality personnel, regulatory, R&D, engineering, manufacturing, packaging, logistics, sales, customer services, finance, legal, and field support. Once this group is assembled, it needs to collaborate to form a detailed list of functional and non-functional requirements, compile a vendor long list, and build and issue an RFP that’s structured for response analysis and comparison.
Evaluation, Scoring, and Selection
Your eventual shortlist vendor demonstrations will typically follow a set format. Your organization should use a standardized scorecard given to each team member with results compiled at the end of the evaluation period. The scorecard should include specific technical capabilities and functionalities as demonstrated by the vendor.
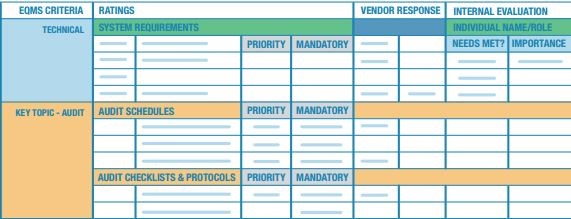
As the scorecard system whittles options down to a true shortlist, your organization should request and follow up on references. A key due diligence component, the goal should be to speak with customers that exhibit similar characteristics to your own, and have implemented the solution within the last 18-24 months or so.
As EQMS implementations become more commonplace across industries, quality organizations that take the time and effort to make these chronological steps find themselves in a better position to achieve early and sustained ROI, as well as the flexibility to integrate additional EQMS functionalities and number of sites as budgets and operational scenarios within their organizations allow.