In February, I had the great opportunity to speak at and participate in QUMAS’s Global User Conference, Connect 2014. Filled with a variety of life sciences professionals, the conference addressed major quality management challenges faced by the industry today. Particularly, much focus was on the rising use and complexity of global partner networks and third-party vendors, and the need for better collaboration capabilities.
In this article, I’ll share some of my key takeaways from the conference. In addition to providing a quick overview of my keynote presentation, I’ll discuss the buzz that was going around about scientific software solutions provider Accelrys’ acquisition of QUMAS. I’ll also share some of my thoughts on QUMAS’s customer case study with Vertex Pharmaceuticals, and how that customer’s requirements were used to drive the release of QUMAS iX.
Keynote: Driving Quality and Compliance Success in the Life Sciences Industry
In the past several years, we’ve spoken to a number of top executives from both life sciences and manufacturing industries. What we’ve found most interesting about those discussions are the varying stages of Quality Management maturity each organization can be categorized within. It’s actually prompted us to develop our new Quality Management Maturity Model.
The model covers six different resource categories, mapping each maturity phase from ad-hoc through market leader.
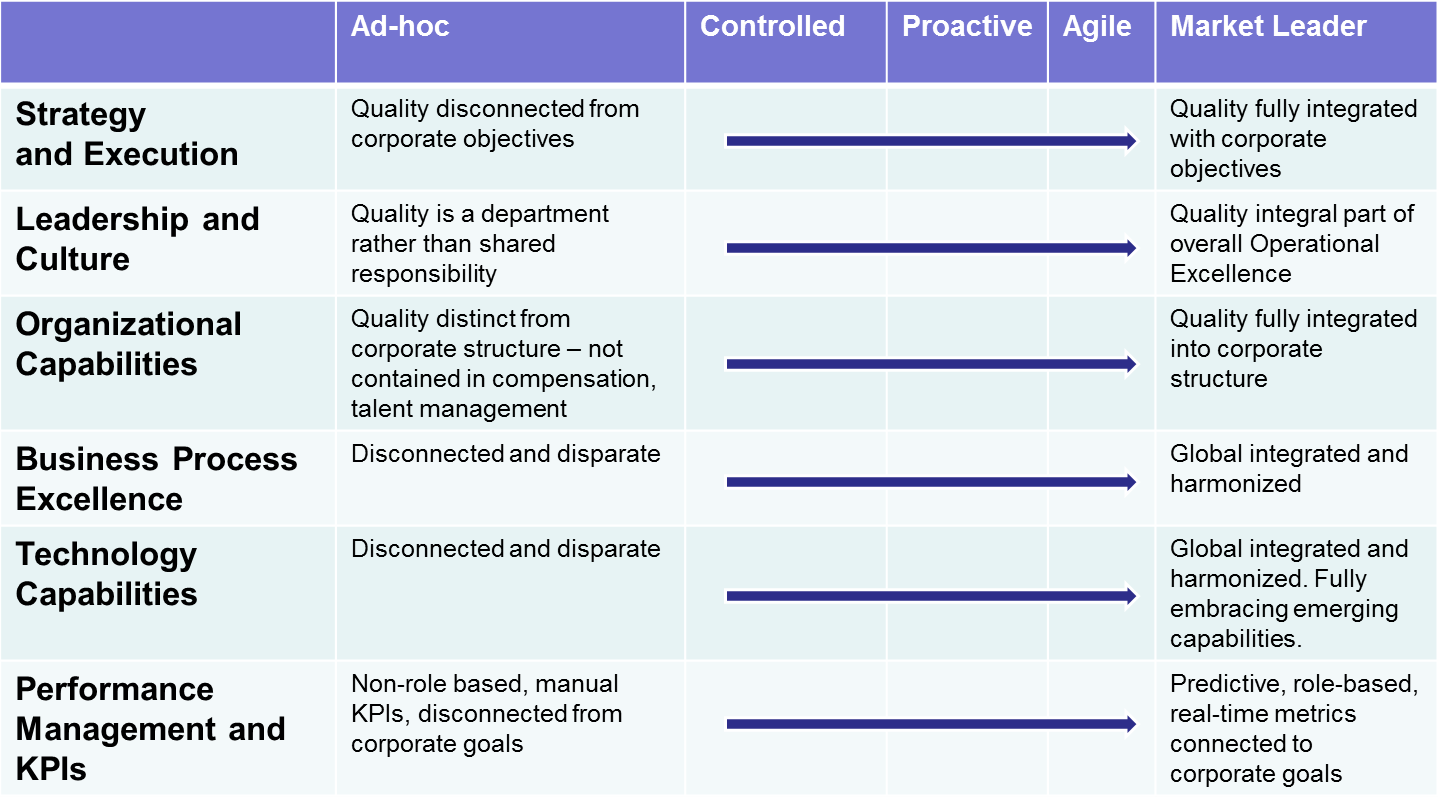
In my presentation, I drilled down into the six different resource categories, sharing specific strategies and best practices for progressing toward market leadership in each. Much focus was on the bottom three categories in the image above: Business Process Excellence, Technology Capabilities, and Performance Management and KPIs.
A short excerpt of the presentation can be found in the SlideShare below, and the whole thing can be found here.
Connecting Scientific Innovation Lifecycle Management and EQMS
Throughout the conference, there was a lot of talk about Accelrys’ acquisition of QUMAS. For those of you who aren’t entirely up to date on the topic, here’s a quick rundown. In December 2013, Enterprise Quality Management Software (EQMS) solutions provider QUMAS was scooped up by scientific software company Accelrys, Inc. for $50 million.
Among a number of other solutions, Accelrys delivers Scientific Innovation Lifecycle Management (SILM) solutions, and saw significant need for and overlay with an EQMS solution like the one offered by QUMAS. The combined solution has added a modernized layer of quality and compliance as well as better collaboration capabilities into Accelrys SILM solution.
Now that we’re all up to date on that acquisition, here’s some more news. In January 2014, Product Lifecycle Management (PLM) giant Dassault Systèmes announced its intent to acquire Accelrys for $750 million. This acquisition will effectively increase Accelrys’ resources and the solution will likely be built into the life sciences arm of Dassault Systèmes.
At LNS Research, we see this movement in the market between QUMAS, Accelrys, and Dassault Systèmes as a positive. Although there will be a lot of moving parts, and it hasn’t been made public exactly how the solutions will be positioned within Dassault Systèmes’ offerings, the connection between PLM, SILM, and EQMS has the potential to truly accelerate innovation and move the space forward.
Vertex Pharmaceuticals’ Collaboration Needs and The Release of QUMAS iX
On the second day of the conference, we heard an interesting case study from Vertex Pharmaceuticals, a biotechnology company that deployed QUMAS DocCompliance roughly eight years ago. The focus of the case study was on collaboration.
For years, Vertex Pharmaceuticals’ personnel were collaborating with external partners and third-party vendors via a combination of traditional methods such as FTP file sharing, email, burned media, and web-based drop boxes. Despite the robust document management deployment already in place for internal operations, there was no easy way to extend that efficiency and capability out to its external partner network.
As this external partner network has only grown in recent years, it was becoming increasingly resource-intensive to manage. Enter QUMAS iX. QUMAS has spent the past several years taking in customer feedback, and has recently released its new web-based solution for securely exchanging and collaborating on controlled regulatory and GxP documents with the external partner network.
Again, we view the release of QUMAS iX as a positive. Like other EQMS vendors serving the life science markets, QUMAS was starting to feel significant pressure from clients to facilitate a secure connection between internal and external operations without an exhausting total cost of ownership. QUMAS has done a good job at listening to its customers, and we will report back on the solution as more use cases come out in the future.
For those interested in learning more about Quality Management Maturity and collaboration in life sciences, take a look at the entire slide deck from my keynote presentation (linked below).