6 Benefits of Moving to a Paperless Manufacturing Environment
LNS Research details six benefits of moving toward a paperless manufacturing shop floor.
Manufacturing and quality management are inextricably connected in process and theory, but often poorly or informally connected in data systems. Manufacturing Operations have deployed Manufacturing Operations Management (MOM) systems to manage manufacturing maintenance, operations and functional quality. Quality Management has deployed an Enterprise Quality Management System (EQMS) to provide overarching quality management, including manufacturing quality governance activities such as Non Conformance (NC) and CAPA. Unfortunately, although both play a vital role in quality management and share certain processes, they are often disconnected systems.
According to LNS Research Principle Analyst Andrew Hughes’ research, manufacturing leaders most commonly identify quality management as the most critical area of MOM software responsible for their company’s success, equal with Production Execution Management. 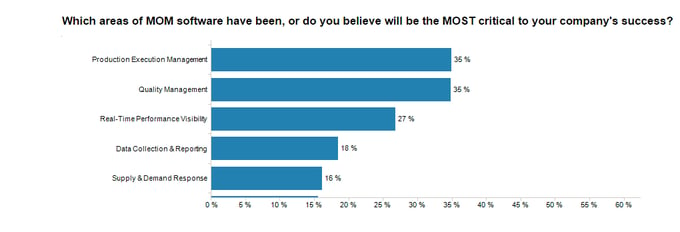
From a quality perspective, those that have implemented closed loop systems across design, manufacturing and suppliers have 48% more successful new product introductions. However, the majority haven’t implemented these yet. Integration of quality systems with other “disparate data sources” is consistently cited as the single top challenge in meeting quality objectives at 38% of respondents.
Often a company may have many MOM or MES systems, as well as hopefully one EQMS. Each MOM will track maintenance, production, quality, and inventory, often at a plant level. Quality data includes processes such as in-line and off-line inspections, Non-Conformances (NCs), as well as possibly CAPAs and SPC. The focus of MOM NCs are on manufacturing, and CAPAs are typically lean, lightweight corrective actions. Referring to the quality Infographic based on the ISA-95 framework, specifically under the make portion of the value chain. MOM primarily operates in the “Functional Quality” portion, although it does have overlap with the “Enterprise Quality” portion.
The EQMS will capture core ISO quality processes such as Audits, Change Management, etc, as well as NC and CAPA. EQMS processes are corporate-wide and as such have tentacles across the corporation. For instance, the NC process in EQMS can be associated with manufacturing, but also Audits and other inputs. Therefore, EQMS primarily operates in the “Enterprise Quality” portion, although because some EQMS solutions support inspection tracking it also dips into the “Functional Quality” portion.
Our recent blog “What System Should Own Our Quality Data?” discussed the need to determine data mastery and traceability, as well as consideration of use scenarios when deciding system overlap. In the case of MOM and EQMS, how should we establish quality information mastery and traceability such that we maximize plant, business unit and corporate quality? Here are some considerations related to:
One of the top challenges in quality management is “disparate systems and data sources.” However, with careful consideration of system interfaces in the interest of maximizing plant, business unit and corporate quality, systems and data sources are no longer disparate and all stakeholders can benefit.
NEW Research Spotlight on strategies and recommendations for minimizing risk through a migration away from monolithic, single-plant MOM architectures through exploration of Cloud and IIoT technologies that are advancing in manufacturing today.
As a member-level partner of LNS Research, you will receive our expert and proven Advisory Services. These exclusive benefits give your team:
Let us help you with key decisions based on our solid research methodology and vast industrial experience.
BOOK A STRATEGY CALLLNS Research details six benefits of moving toward a paperless manufacturing shop floor.
Learn why leading Life Sciences companies are focusing more on the connection between manufacturing operations and quality management.
LNS Research discusses its 2013 expansion into Manufacturing Operations Management and Sustainability.
The Industrial Transformation and Operational Excellence Blog is an informal environment for our analysts to share thoughts and insights on a range of technology and business topics.