The life sciences industry’s regulatory environment has both encouraged and mandated companies to adopt today’s emerging quality management technologies. Particularly around areas such as risk, compliance, corrective and preventive actions, employee training, supplier quality management, document management, and product traceability, there’s great need for process automation and harmonization. These consumer-protecting, risk-mitigating functionalities can be found in most modern Enterprise Quality Management Software (EQMS) solutions.
One EQMS vendor that’s made a name for itself in the life sciences industry is MasterControl. Transforming from a document control software company to a provider of enterprise quality software and solutions in the past 20 years, MasterControl’s strong set of functionalities is serving a range of operations around the globe. In this article, we’ll inspect data on the adoption of EQMS solutions in life sciences, and dive into the inner-workings of MasterControl’s EQMS capabilities.
The Rise of Enterprise Quality Management Software in Life Sciences
From a recent quality management survey conducted by LNS Research, it’s evident that adoption of EQMS solutions is on the rise. The survey asked more than 500 executives across all industries about their quality management technology adoption and 20% reported already having some type of EQMS solution in place. While this number is a good indicator of the market, what’s more interesting is that almost 30% of all executives reported that their organization planned to adopt an EQMS solution in the near future.
And, looking solely at the life sciences industry, impressively, 33% of companies are in the planning stages.
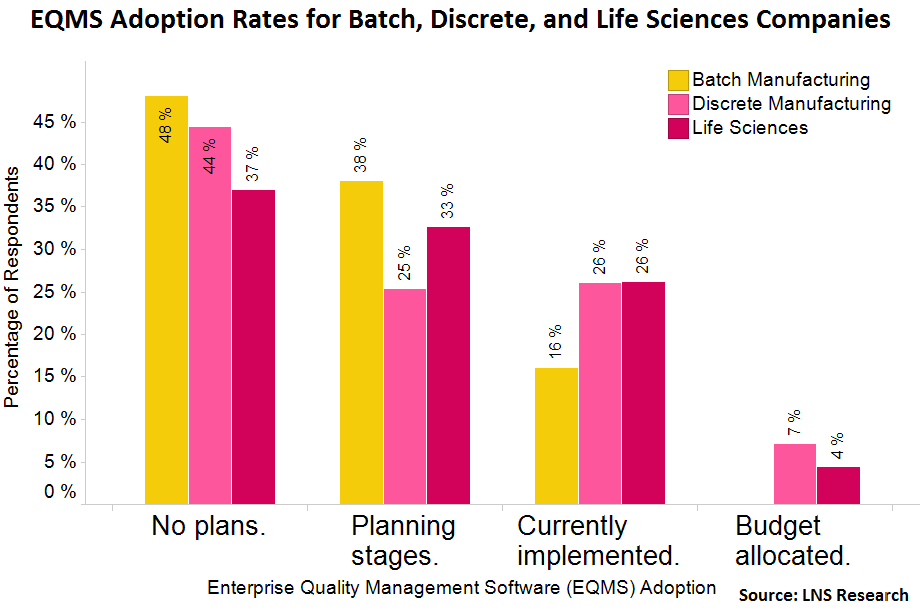
In our calls with senior leaders of some of the world’s largest life sciences companies, EQMS is becoming more and more of a focal point. Among others in the space, MasterControl is one name that’s often discussed.
MasterControl’s Quality Management Solution
Founded in 1993 and based out of Salt Lake City, UT, MasterControl started as a document control software provider. Since then, the company has evolved into one of the leading providers of Enterprise Quality Management Software solutions with over 500 customers. Its EQMS solution has been widely deployed across the life sciences industries—particularly in pharmaceutical, medical devices, and blood and biologics—in addition to other highly regulated industries.
MasterControl serves a range of company sizes, but focuses on enterprise-wide implementations for larger, global operations. In contrast to some EQMS solutions providers, MasterControl has the scale to serve a global customer base, with service and implementation employees distributed across all major regions. Additionally, MasterControl uses its strong employee base to conduct professional training courses for both customers and non-customers in these regions.
Covering a wide range of functionality, MasterControl’s EQMS suite encompasses capabilities such as quality event management, documents management, corrective and preventive action management, audit management, supplier quality management, risk management, employee training, and more. It also offers complementary solutions for areas including PLM (PDM links, BoM, etc.), LIMS, MES, and ERP, as well as for connecting with customers, suppliers, and partners for pharma-regulatory control purposes.
Although MasterControl originated in the document management software space with a traditional client server offering, it launched its Web-based process-centric platform in 2004. Since the launch, MasterControl has made significant investments to build out its platform as a hub for quality functionality. With nearly 70 different functionalities today, customers can deploy each individually and scale incrementally, or follow a more pervasive deployment model and leverage MasterControl’s entire suite. A large majority of customers deploy more than one product today.
MasterControl deployed its first cloud-based solution in 2007 and over the past six years; the solution provider’s seen an increasing adoption of its cloud-based offering. While it’s not a true multi-tenant solution (neither are other EQMS vendors), the company chose to follow a hybrid model because of the heavily regulated environment. However, based on customer preference there is flexibility to have a completely segmented solution. In the future, we expect to see the solutions provider invest more in its cloud-based offering.
Additionally, we expect to see MasterControl move deeper into supplier quality management for tier 1 pharmaceutical manufacturing companies as well as offer more out-of-the-box functionality for the life sciences industry as a whole. Other investments LNS hopes to see in the coming years from MasterControl involve increasing focus on mobility and analytics capabilities, keeping pace with trends in the market toward quality as a driver of continuous improvement.
Considering a MasterControl Implementation?
LNS will have additional coverage of the vendor’s platform improvements and investments in the updated 2014 version of the EQMS Solution Selection Guide. If you're considering MasterControl or another quality management software solution provider for your next implementation and would like insight from LNS Research, you can download the report below and/or contact us direclty at matthew.littlefield@lnsresearch.com.
