In mid-2017, LNS Research identified that an early but significant group of manufacturers had begun to recognize Quality 4.0 as one of their top Industrial Transformation initiatives. LNS coined the term Quality 4.0 and defined the space - why to adopt Quality 4.0, and how to build a successful strategy. Over time, the interest in Quality 4.0 has continued to accelerate, and many manufacturers are now either investigating Quality 4.0 or deploying early pilots.
Putting this in the parlance of Geoffrey Moore’s famous publication “Crossing the Chasm,” this wave of manufacturers from mid-2017 onward are “early adopters,” and follow the Quality 4.0 “innovators,” who are now on their 2nd or 3rd generations use cases. Moore expected that innovators and early adopters would encompass 16% of the market. Interestingly, this aligns well with LNS’ statistics, which identify that currently, 13.8% of the market has already begun its Quality 4.0 journey.
Pharmaceutical firms have been one of the early adopters of Quality 4.0 and have already deployed Quality 4.0 use cases across product development, clinical trials, laboratories, manufacturing operations, logistics, and pharmacovigilance. Many are pivoting to explore opportunities presented by the FDA’s increasing openness to real-world data while finding solutions that improve the ability to navigate global compliance complexity.
Learn from Early Mistakes
Quality 4.0 innovators experienced successes and setbacks in their journeys. Many in the broad market have been trapped in what is being called “pilot purgatory,” where initial Industrial Transformation pilots do not either deliver value or scale.
Several root causes are prevalent in these early failures:
Shiny Object Syndrome (SOS):
One prevalent idea is that firms can deploy new tech in many separate pilots without clearly defined business value. Business value is not an incidental byproduct; start with strategic/peer objectives and tightly define use cases.
Tech vs. Transformation:
While technology is the enabler, it is not the end goal. Those that do not transform their cultures, products, and processes spend substantial financial and time outlay to achieve marginal results (or churn). LNS’ core Quality 4.0 research highlights the importance to transform across 11 axes.
New Digital Silos:
Many firms have independent initiatives that have sprung up at various sites and across multiple parts of the value chain, in support of the “fail fast” mantra. These initiatives do not build upon each other and are creating new digital silos to replace traditional silos. By contrast, successful firms have typically taken a centralized, flexible approach that ultimately integrates and scales transformation.
Lack of Top Management Support:
As with most transformations, a bottom-up or mid-out transformation is much slower and less effective than one that can rely on top management to softly or firmly encourage adoption. LNS’ quality maturity research identifies that quality teams with top management priority have 2.9 times more people, process, and tech capabilities than those without. This helps quantify top management’s impact on success.
Site Readiness:
Larger firms are aware that some sites have a much greater appetite and an ability for change than others. Also, in some firms, the power may rest with large sites or business units rather than corporate. While LNS is exploring this in more detail in forthcoming research, the advice, for now, is to be careful to identify the sites that are most ready and willing to transform and use these sites as beacons of success.
Poor Solution Selection:
Solution selection is where many of the above bad behaviors are embodied. It also is where teams often waste precious months or years investigating solutions without realizing that they are squandering time to value.
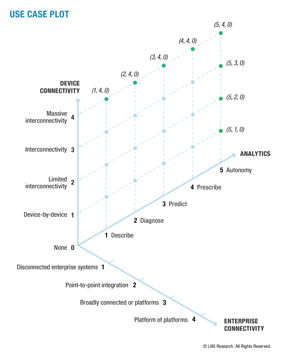 The Path Forward
The Path Forward
LNS is excited to launch the concept of “use case plots.” Use case plots are built to address many of the challenges outlined in this blog. They make it is possible to map the capabilities deployed in the operational theatre and in the enterprise software theatre, with analytics and insights.
The use case plots are then used to map a journey from the current state to a transformative future state. For instance, pharmaceutical firms can leverage use case plots to grow from a low-capability environment to individually connected manufacturing assets to predictive product quality, then from predictive product quality to predictive patient experiences.
Pharmaceutical firms are increasingly adopting technology to drive Quality 4.0. Firms are investigating this journey, and those currently underway should get educated on how others have succeeded and failed on their road to Quality 4.0.