Approaches to Quality Management have changed dramatically in the last decade. Some industrial companies still struggle with paper-based quality approaches. Meanwhile, others are stitching together a homegrown version of Quality Management capabilities by hobbling together point solutions not intended as Quality Management solutions. The Enterprise Quality Management Software (EQMS) vendor landscape is crowded and growing more complex each year, with several types of platforms and focus areas.
An EQMS is a key element of a Collaborative Quality strategy. Its primary role is facilitating cross-functional communication and collaboration across the value chain by centralizing, streamlining, and standardizing quality processes and data from traditionally disconnected business processes. Interoperable "System of Systems" have extended Quality's influence into partner functions.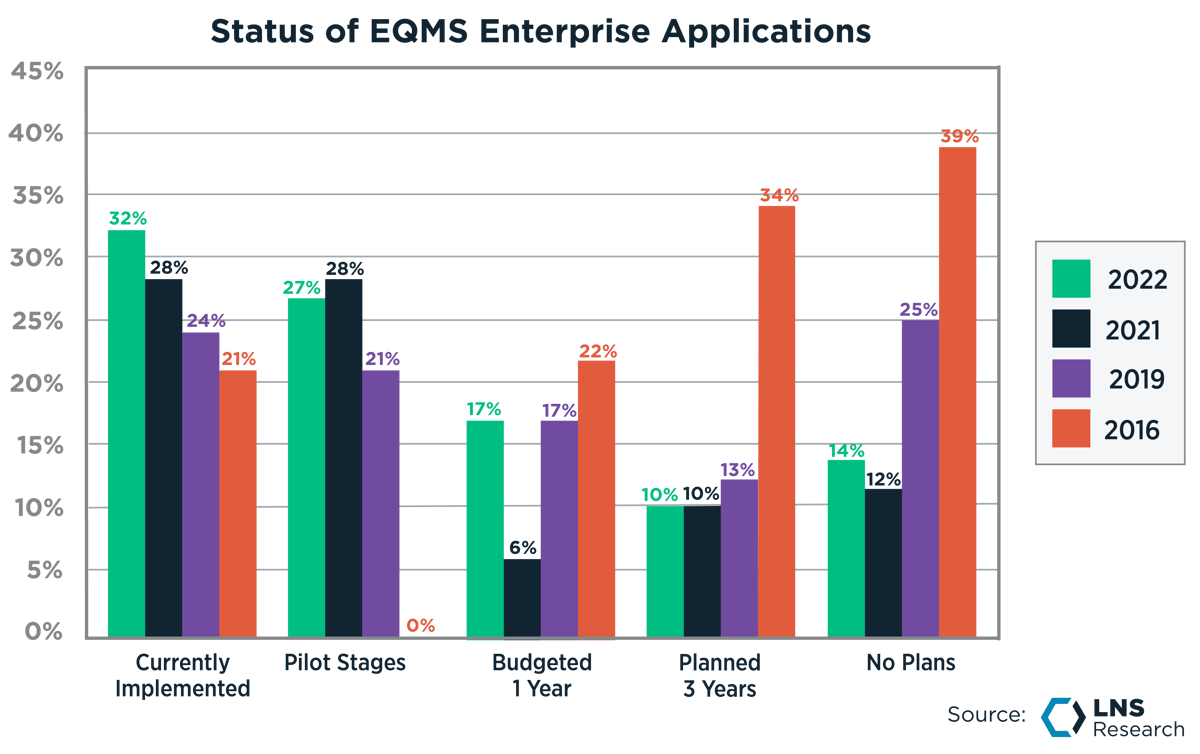
In recent years, we have seen a shift in the adoption of EQMS over no decision, which had previously been the top competitor in EQMS decision-making. Now, we see EQMS becoming an accepted software that Quality leaders should have to manage their responsibilities effectively. Fifty-nine percent of industrial companies have either implemented or are piloting an EQMS.
Advancements in this space have been rapid, and vendors from beyond just Quality have invested in Quality Management functionalities. Consequently, there are many options for companies interested in an implementation, from tangential solutions with EQMS functionalities to Stand-alone EQMS.
Below, I'll discuss the different types of implementations available to users today, providing insight on each and potential vendors to look into.
You Have Many Options in EQMS
-
-
-
Stand-alone Enterprise Quality Management Software
EQMS delivers the capability to manage the quality of products and processes across the value chain from a single location. By integrating EQMS functionalities such as CAPA management, risk management, audit management, and supplier quality management with SCM, CRM, ERP, PLM, and more, executives can have an unparalleled viewpoint of global quality. .png?width=582&height=632&name=2023%20Enterprise%20Quality%20Management%20Software%20(EQMS).png)
With the maturity of the pure-play market and vendor experience with enterprise-wide rollouts, there are several strong options to consider when selecting an EQMS. Some of these include EtQ, Intellect, ComplianceQuest, and AlisQI, just to name a few. While these vendors started with smaller site implementations, some are now adding enterprise-wide solutions with robust compliance and process management capabilities.
EQMS is moving closer to the vision of Collaborative Quality, which creates positive impacts across the value chain with more cross-functional capabilities and incorporation of large data sources, not just event management but also manufacturing and supplier processes.
-
Product Lifecycle Management-based EQMS Solutions
PLM-based Quality software has helped many engineering-intensive organizations improve the quality of products and processes. While these solutions alone are not as robust as those offered in the pure-play EQMS market, companies see value in having a single solution to manage engineering and quality processes. It is also typical for PLM solutions to be costly, which tends to take away from the Quality Management software budget. However, more vendors in this space are now investing in Quality-specific functionalities such as CAPA, and we expect these solutions to continue to improve.
Since a full EQMS implementation may not be feasible for engineering-intensive companies, many PLM vendors have developed EQMS functionalities. There are big brand names in this category. Companies such as Siemens, PTC, and Dassault Systèmes have a solid presence in the PLM space and have also developed Quality Management solutions that extend from engineering to Quality functions.
-
Enterprise Resource Planning-based Quality Management Software
Companies have spent more money and resources implementing ERP solutions than any other available solution on the market. ERP is generally the IT backbone of organizations, carrying out many functions under a single system. With that said, it has only been a natural progression for ERP vendors to develop and invest in quality management functionalities. A recent example of this is QAD's acquisition of CEBOS.
The most significant advantage of leveraging Quality Management functionalities in ERP is having a single enterprise solution for managing cross-functional processes. Like the PLM space, though, pricing can be an issue, and ERP Quality Management solutions are not necessarily as robust as the portfolio of functionalities offered by a pure-play EQMS vendor. The benefit of investing in ERP-based Quality solutions is not having to manage multiple-point solutions to manage different organizational processes. Vendors to look into include Rockwell (Plex Systems), Oracle, QAD, and SAP.
-
Integrated Suites EQMS Solutions
The EQMS Market has splintered into general solutions, those mentioned above, and industry-specific solutions with some unique features that are requirements of that industry. The greatest bifurcation has occurred around Life Science EQMS solutions. These EQMS solutions have all the typical functionality of an EQMS in general, with some additional features and functions to help users address the typical pain points unique to a regulated industry. Features may include Statistical Process Control, traceability into supplier quality, event management, investigation functions, and clinical trial management functionality in some cases. MasterControl, IQVIA, Veeva Systems' Life Science Cloud, and Honeywell Connected Life Sciences are some vendors to consider in this category.
Recommendations for Quality Leaders
 Evaluate strategies aligned with your business model. If your organization is an Engineering/R&D focused company, consider solutions that flow from design and PLM solution providers. Consider solutions from pure-play or ERP-based providers if your company is an Operations or Supply Chain-focused company with a low or steady new product burden.
Evaluate strategies aligned with your business model. If your organization is an Engineering/R&D focused company, consider solutions that flow from design and PLM solution providers. Consider solutions from pure-play or ERP-based providers if your company is an Operations or Supply Chain-focused company with a low or steady new product burden.
 Choose the most flexible solution if you serve multiple industry sectors. Many industrial companies provide products in several industry sectors, some regulated, some not. Industries, such as Life Sciences and Aerospace have higher regulatory burdens. Automotive or Industrial Equipment is less so. Varying degrees of regulatory requirement dictates choosing the solution with the features needed to support the most stringent regulatory requirement but with the flexibility to allow other parts of the business to use a less rigorous version.
Choose the most flexible solution if you serve multiple industry sectors. Many industrial companies provide products in several industry sectors, some regulated, some not. Industries, such as Life Sciences and Aerospace have higher regulatory burdens. Automotive or Industrial Equipment is less so. Varying degrees of regulatory requirement dictates choosing the solution with the features needed to support the most stringent regulatory requirement but with the flexibility to allow other parts of the business to use a less rigorous version.
 Determine business drivers and user personas to prioritize. Many EQMS offerings have multitudes of embedded out-of-the-box applications. Think strategically about where to start, but don't stop at the four walls of the Quality function. Significant performance improvements can be gained by integrating an EQMS into a System of Systems approach of interoperable software across the enterprise.
Determine business drivers and user personas to prioritize. Many EQMS offerings have multitudes of embedded out-of-the-box applications. Think strategically about where to start, but don't stop at the four walls of the Quality function. Significant performance improvements can be gained by integrating an EQMS into a System of Systems approach of interoperable software across the enterprise.
 Future-proof your strategy. While the scope of the initial deployment should focus on the current challenges to be addressed, anticipate future needs so that a " future-proof " system is selected to avoid having to rip and replace later when additional capabilities are needed.
Future-proof your strategy. While the scope of the initial deployment should focus on the current challenges to be addressed, anticipate future needs so that a " future-proof " system is selected to avoid having to rip and replace later when additional capabilities are needed.
Above all, Educate Yourself.
The decision often ends with the consensus of a cross-functional team. However, it typically begins with some personal research. The LNS Research EQMS Solution Selection Matrix and Guidebook has helped hundreds of executives with their EQMS journey and is being refreshed for release in 2024. Make it the first stop on your way by following the link below.
Related Resource References:
